Can you make psilocybin drops from truffles or magic mushrooms? I would like to make the mushroom or truffle drops myself so I know for sure what is in them. Do you have any tips?
Easiest way to make mushroom drops with ethanol
In theory, the following alcohol extraction method by evaporation is one of the least complex and relatively safe ways that you could do at home. This is a step-by-step overview of an alcohol extraction with natural evaporation, where the temperature remains low to prevent the degradation of psilocybin.
Supplies:
- Dried psilocybin-containing mushrooms or truffles (undried is also possible, but then there is more water involved in the entire process)
- Ethanol ≥ 95% (preferably potable alcohol such as Everclear or pharmaceutical grade)
- Mill or grinder
- Dark glass jar or bottle
- Coffee filter or fine cloth
- Evaporator tray (e.g. glass oven dish)
- Dark glass bottle with dropper
- Lemon juice or citric acid (optional, for stability)
- Gloves, well-ventilated room
Steps
1. Grinding the material
Grind the dried material into a fine powder. The finer the powder, the more efficient the extraction. However, do not expose it to oxygen for too long.
2. Preserving (maceration) in ethanol
Place the powder in a sealable jar. Add ethanol in a ratio of 1 part powder to 10–20 parts alcohol (v/v). Optionally, add a splash of lemon juice or natural vinegar to make the solution slightly acidic.
Let this Steep for at least 24 hours in a cool, dark place. Shaking gently occasionally helps.
3. Filter
After soaking, filter the liquid through a coffee filter or cheesecloth. This separates the mushroom material from the liquid extract.
4. Slow evaporation
Pour the filtered liquid into a wide, open glass bowl. Let it at room temperature (not above 30–35°C!) evaporate in a dark, well-ventilated place. This takes days to weeks, depending on the amount of alcohol and the surface area. It is therefore important to create as large a surface area as possible.
Caution: DO NOT heat on a stovetop or in an oven. Alcohol is highly flammable. Use only natural evaporation.
You can stop vaporizing as soon as you reach the desired concentration: e.g., 10 ml extract with X mg psilocybin. Some people let it thicken into an almost syrupy liquid for dosing with a pipette.
5. Transfer and store
Pour the concentrated liquid into a dark bottle with a dropper. Store in a cool, dark place. If you maintain an alcohol percentage above 20%, it will remain usable for longer (months to years).
Important points to consider
- Psilocybin is reasonably heat resistant, but not psilocin. So keep the process cold or lukewarm.
- No open flames, heaters, or smoking during alcohol evaporation. Fire hazard!
- Alcohol evaporates poorly at low temperatures. It takes a long time, be patient.
- This method yields a raw extract, not purified psilocybin. Dosage remains an estimate unless you have it analyzed.
- Legal status: In the Netherlands, you are not allowed to extract psilocybin or psilocin outside of scientific research.
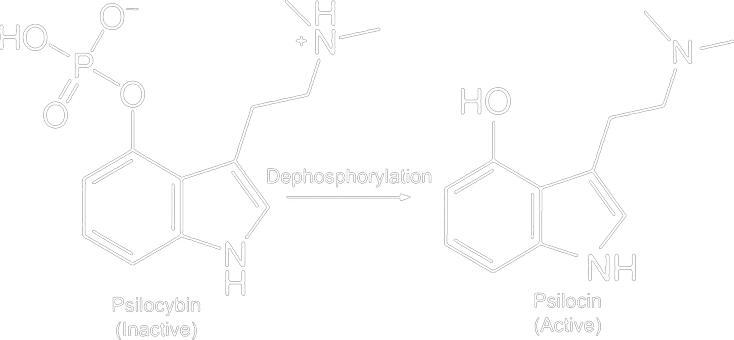
In the Netherlands, it is prohibited to possess psilocybin and psilocin without an exemption (for scientific research). Extracting psilocybin from legal truffles is therefore prohibited and is only permitted if you have an exemption for it.
The best way to extract as much psilocybin and psilocin as possible from truffles or magic mushrooms is an ultrasonic extraction process (“ultrasonic bath extraction”) in combination with polar solvents (methanol or methanol-water mixtures).
The extraction principle
Psilocybin Both methanol and psilocin are polar. Therefore, they dissolve best in a polar solvent such as methanol, methanol-water (e.g., 80:20), or ethanol-water. If you are concerned about solvent residue, choose ethanol over methanol (ethanol is the alcohol we know from spirits, and methanol is the highly toxic, dangerous alcohol).
The reason an ultrasonic bath works so well is because sound waves create microbubbles in the liquid (cavitation). These burst against the plant/fungal tissue, causing the cell walls to break and the active ingredients to be released faster and more completely.
The method with the highest yield
If you want to extract the most active ingredients from the material, then the optimal working methods are:
1. Use dried material
- Dried mushrooms or truffles are finely ground into powder.
- This increases the surface area and makes extraction more efficient.
2. Polar solvent (usually methanol + water)
The highest psilocybin yield is achieved with:
- Methanol
- Methanol with a little water (80–95% MeOH)
- Safer alternative with almost the same yield: Ethanol (80%-95%)
Why methanol?
It has the best balance between polarity and penetration power for psilocybin.
3. High solvent-to-sample ratio
- Minimum 1:100 (m/f)
For example, 1 gram of dried material per 100 ml of solvent.
This prevents saturation of the solution and promotes complete extraction.
4. Ultrasonic extraction (ultrasonic bath)
- 20–40 °C (preferably around 20–25 °C to prevent degradation of psilocin)
- 30–60 minutes ultrasound treatment
- Cavitation breaks open cell walls → active substances are released
5. Multiple extraction
Instead of extracting once, the same material is extracted more often:
- Add solvent
- Ultrasonic bath
- Filter
- Add new solvent
- Ultrasonic extraction once again
The liquids are combined → higher total yield (although each subsequent extraction has a lower yield).
6. Low temperature
The temperature must remain low because psilocin degrades rapidly in the presence of heat or oxygen.
Psilocybin is more stable, but lower temperatures yield better overall results.
7. Filtration and evaporation
The solution is filtered to separate solid particles.
Subsequently, the solvent is carefully removed. This can be done using a rotary evaporator (low temperature and vacuum) or by heating the liquid (heat also results in a loss of active ingredients).
After this, you are left with a mixture of active ingredients, water, and possibly a small residue of the solvent (note that with methanol, complete evaporation is required; ethanol is safer and is better to use).
Just a quick note on the choice between methanol and ethanol:
Methanol has a much lower boiling point than ethanol (64.7 °C versus 78.37 °C). This means that methanol vaporizes at a relatively low temperature. Therefore, when heating an extract to evaporate the solvent, methanol reaches its boiling point much faster than ethanol.
Result:
- You don't need the liquid to heat less intensely to evaporate methanol.
- The extract is therefore shorter exposure to heat, causing sensitive substances (such as psilocin) to degrade less rapidly.
- The total thermal load of the extract is much lower.
Theoretically, therefore, you can remove methanol faster and more efficiently by moderate heating, because the temperature required to reach the boiling point just under the critical threshold remains at which psilocin begins to break down significantly.
Why ethanol actually causes more damage due to its higher boiling point
Ethanol only evaporates at 78+ °C, and that is a problem for sensitive tryptamines.
The degradation of psilocin – and to a lesser extent psilocybin – accelerates sharply above 70 °C, and with prolonged heating even starting from 60 °C.
That means that:
-
To completely remove ethanol, you must heat the extract and keep it warm for longer.
-
That higher temperature falls within the range where psilocin oxidizes and breaks down rapidly.
-
The longer duration above this critical limit causes substantial losses of active ingredients.
In other words:
Ethanol is safer as a solvent, but less safe for the active ingredients during evaporation because you have to heat the mixture much more and thereby lose valuable compounds.
The core difference explained logically
Methanol → low boiling point → little heat required → less degradation
Ethanol → high boiling point → more heat needed → more degradation
The solvents therefore differ not only in toxicity, but especially in how strongly they raise the temperature of the extract during evaporation. Methanol requires much less energy and time, whereas ethanol keeps extracts for too long in a temperature zone where psilocin is unstable.
Psilocybin is the main psychoactive substance in so-called “magic mushrooms” and truffles, and is converted in the body into the active compound psilocin. A liquid extract in droplet form (similar to a tincture) can offer advantages: dosage becomes more precisely controllable and one does not have to ingest the sometimes unpleasant material of the mushroom itself. This study discusses whether it is chemically feasible to extract psilocybin from mushrooms or truffles into a standardized liquid. We cover suitable extraction techniques, the use of solvents (such as alcohol), the stability of psilocybin in liquid form, and the theoretical dosing possibilities of such an extract. The process is described below. theoretical and educational described, without practical instructions for use.
Chemical feasibility of a psilocybin extract
Chemically speaking, it is possible to isolate psilocybin from mushrooms in a concentrated solution. Psilocybin is readily soluble in water and polar solvents. In contrast, pure psilocybin is only with difficulty soluble in pure ethanol – therefore, mixtures of water and alcohol are often used to ensure sufficient solubility. Also fresh truffles (sclerotia) contain psilocybin and psilocin and can theoretically be processed in the same way.
The distinction between is important psilocybin and psilocin. Psilocybin (a phosphate ester) is chemically more stable than psilocin. Psilocin is more susceptible to degradation (oxidation) when exposed to oxygen, light, or heat. Therefore, during extraction, one ideally wants to preserve as much of the stable psilocybin as possible. In the natural material, psilocybin is usually present in larger quantities than psilocin, partly due to this higher stability. An extract from dried mushrooms will therefore contain primarily psilocybin, which is beneficial for shelf life. The psilocybin can later (upon consumption) be converted into psilocin in the body, which causes the psychoactive effects.
Suitable extraction techniques
Various extraction methods are conceivable for the theoretical obtaining of a psilocybin-containing extract. Some important examples are:
-
Maceration (weeks)Finely ground dried mushrooms or truffles are soaked for an extended period at room temperature in a solvent (e.g., a water/alcohol mixture). This simple method gradually extracts the alkaloids from the plant material. A sufficiently long soaking time (at least 30 minutes to several hours) is often recommended for optimal yield.
-
Reflux/Soxhlet extraction: In this process, the solvent is continuously circulated through the heated mushroom material. This intensifies the extraction, but one must be careful with heat handle to avoid degradation of psilocybin. Excessively high temperatures or strongly alkaline/acidic conditions can hydrolyze psilocybin into psilocin, which can subsequently oxidize and become inactive. Therefore, extraction is often performed at moderate temperatures or for a short duration.
-
Ultrasonic extractionUltrasonic sound (as applied in an ultrasonic bath or probe) greatly accelerates extraction. It vibrates the cell walls of the mushrooms, causing the active substances to be released rapidly. Cold ultrasonic extraction (e.g., in ice water) can yield a high return of psilocybin and related alkaloids within minutes. This method provides very high yields and works efficiently at low temperatures, which minimizes the degradation of heat-sensitive substances.
-
Other methods: Advanced techniques have also been described in laboratories, such as supercritical fluid extraction (e.g. with CO₂) or solid-phase extraction. Although such methods can potentially yield a very pure extract, they require specialized equipment and are more relevant on an industrial or pharmaceutical scale than for small-scale application. Furthermore, after crude extraction, further Purification it is possible (e.g. via filtration, solvent extractions, or chromatography) to obtain nearly pure psilocybin crystals. However, this is complex and requires extensive chemical knowledge, and falls outside the scope of a simple “droplet” extract.
In theory, a simple homemade tincture-like extract (such as maceration or ultrasound) would suffice to dissolve psilocybin. Complex purification steps are not necessarily required unless one wishes to produce pure crystalline psilocybin. The most important thing is that the chosen method efficiently extracts the psilocybin from the mushroom material without causing excessive degradation.
Use of solvents (such as alcohol)
Polar solvents appear to be the most suitable for extracting psilocybin, because the molecule is quite polar (thanks to the phosphate group). Commonly used solvents are:
-
Ethanol (Drinkable alcohol): One of the most commonly used solvents for psilocybin extraction. Ethanol is relatively safe to use and can dissolve psilocybin well, especially when mixed with a little water. In practice, a mixture of water and ethanol is often used to efficiently extract both polar (water-soluble) and slightly less polar substances. After extraction, the alcohol-water mixture can be (partially) evaporated to obtain a more concentrated extract.
-
WaterWater alone can also dissolve psilocybin, especially warm water. Mushrooms, for example, are often used as tea extracted – a hot-water extract – whereby psilocybin ends up in the tea water. Water has the advantage of being non-toxic; however, spoilage must be taken into account (see stability below). Moreover, pure water extractions may be somewhat less complete than alcoholic ones, but with the addition of an acid (e.g. lemon juice) they become more effective. In this way, psilocybin can be ultrasonically extracted with cold or hot water.
-
Other alcohols: Isopropanol (isopropyl alcohol) and methanol There are also solvents that can dissolve psilocybin well. These are regularly used in research. Methanol often yields a high return, especially when acidified, but is highly toxic and suitable only for laboratory purposes. Isopropanol is less toxic than methanol but is not intended for consumption; residues must evaporate completely. For this reason, ethanol is preferred for consumption purposes.
-
Acetone: This organic solvent can dissolve psilocybin to some extent, but is rarely used due to its flammability and toxicity.
-
Acidic additivesAdding a mild acid to the solvent is a well-known strategy. Citric acid (lemon juice) or acetic acid (vinegar) lowers the pH, which offers two benefits. First, a low pH can increase the solubility of psilocybin/psilocin and potentially keep the phosphate group better in solution. Second, acid protects the unstable psilocin from oxidation. An example is the so-called “Lemon Tek”, in which finely ground mushrooms are soaked in lemon juice; this quickly extracts the alkaloids, and the acidic environment prevents psilocin (partially formed from psilocybin in an acidic environment) from breaking down before ingestion. For a shelf-stable extract, citric acid or ascorbic acid (vitamin C) is a useful addition: these antioxidants act as a preservative and stabilizer.
In summary, a mixture of water and ethanol will be ideal as a solvent in many cases. This combines the extraction power of water for psilocybin (which dissolves very well in water) with the preservative and solubility-enhancing properties of ethanol. In technical literature, for example, methanol with a little acid is mentioned as optimal (high yield, low degradation) solvent – for practical purposes, ethanol with water and optionally a dash of acid is the more accessible counterpart.
Stability of psilocybin in liquid form
Stability is a crucial factor in a psilocybin solution. In dry form in an intact mushroom, psilocybin is fairly stable, but once dissolved or isolated, several points must be taken into consideration:
-
Protection against degradationPsilocybin is reasonably stable, but psilocin degrades easily under unfavorable conditions. During extraction, some psilocybin can inevitably be converted into psilocin (for example, by enzymes or heat once the mushroom cells break). This psilocin can then rapidly oxidize upon contact with oxygen into blue-black inactive products (the phenomenon that also causes bruised mushrooms to turn blue). To minimize this, extraction is preferably kept to a minimum. cool and oxygen-poor. In practical terms, this means: avoid high temperatures (preferably cold/room temperature), protect the extract from air (store in a sealed container), and optionally add antioxidants. A slightly acidic environment (pH ~4) is recommended, as this stabilizes psilocin against oxidation.
-
Alcohol content and spoilageA liquid extract is subject to microbial growth if it contains nutrients. Mushroom extract contains mainly alkaloids (no sugars or food), but if the solvent is pure water, mold or bacterial growth can still occur over time. Alcohol acts as a preservative: an ethanol percentage of around 20–25% vol. is usually sufficient for the extract shelf-stable (shelf life at room temperature) to make. At that alcohol percentage, microorganisms cannot grow well. If the alcohol content is lower (e.g. in an aqueous extract without sufficient ethanol), the extract must be stored in the refrigerator and used within a few days to weeks to prevent spoilage or fermentation.
-
Light and temperature: Like many organic compounds, psilocybin degrades more rapidly under the influence of UV light and heat. Therefore, a psilocybin solution is ideally stored in the dark (dark bottle) and at a low temperature. In theory, storage in the freezer one of the best ways to preserve potency for a long time. Frozen or very cold strongly slows down chemical degradation. (Alcohol-water mixtures of ~25% ethanol do not freeze completely hard at -20°C and remain liquid, which can be useful.)
Due to these precautions, a psilocybin-containing liquid can remain quite stable. Under research conditions, for example, it has been found that dried mushroom powder could lose approximately 50% of its psilocybin over several months at room temperature exposed to light. A correctly prepared and stored extract, on the other hand—dark, cool, sufficient alcohol, slightly acidic—should retain its efficacy much longer. Manufacturers of psilocybin tinctures therefore sometimes also add preservatives (such as citric acid or sorbic acid) to further increase shelf life.
Conclusion regarding stability: Psilocybin in a good solution is reasonably stable, but the solution must be prepared and stored properly to prevent loss of potency and microbial growth. Theoretically, with the right approach, a liquid extract can remain effective for many months without significant degradation.
Theoretical dosing possibilities
An important reason to want psilocybin in droplet form is the able to dose accurately. In raw mushrooms, the content varies per specimen, but in a homogeneous extract, the dose per milliliter can be known precisely. Theoretically, the total psilocybin content of the extract is determined by the amount of starting material and the extraction efficiency. Suppose, for example, that ~100 mg of psilocybin is extracted from 10 grams of dried mushrooms (for illustration, at ~1% content). If this is dissolved in 100 ml of liquid, every 1 ml contains approximately 1 mg of psilocybin. Using a dropper (typically ~20 drops per ml), each drop would then deliver approximately 0.05 mg of psilocybin. Naturally, the extract can also be made much more concentrated – some commercial products, for example, offer 30 doses in a small bottle, where each drop or pipette dose represents a microdosing.
In practice, psilocybin doses are often categorized as follows: a microdosing is less than ~2 mg of pure psilocybin (below the perception threshold), an average psychedelic dose is about 10–20 mg, and a high dose can reach 30–40 mg. For comparison: 1 to 3.5 grams of dried mushrooms corresponds to approximately 10–50 mg of psilocybin. With a standardized liquid extract, one can fine-tune very finely within this range. One could, for example, create a formula where one drop contains a sub-perceptual microdose (e.g. 0.5–1 mg), or conversely such that a full pipette (usually ~1 ml, ~20 drops) delivers a full trip dose of roughly 15–20 mg. This depends on the concentration of psilocybin in the solution.
It is important that the solution is well mixed and homogenized so that every drop has the same concentration. In a pharmaceutical context, a standardization step is therefore often performed after extraction: the concentrated extract is diluted to an exactly known strength. Such a standardized extract can then be dosed in, for example, milliliters or drops. In clinical studies with psilocybin, liquid formulations or capsules with precisely weighed amounts are also used (often 25 mg for a therapy session).
Theoretical example: If 100 mg of psilocybin is dissolved in 10 ml of solution, one has 10 mg per ml. A typical drop (~0.05 ml) then contains ~0.5 mg of psilocybin. To ingest ~10 mg, one would need approximately 20 drops (which amounts to one full 1 ml pipette). For a light dose of ~5 mg, ~10 drops suffice, while for a microdose of ~1 mg, only 2 drops are needed. Naturally, the ratio can be adjusted – by making a more concentrated extract, the number of drops per dose can be reduced. Manufacturers of psilocybin microdosing drops offer, for example, bottles where each dose (e.g., 1–2 drops) corresponds to 0.125 grams of dried mushrooms, which amounts to approximately 1–2 mg of psilocybin per dose (a typical microdose). This illustrates that a dropper form is not only feasible but is already actually being used to enable precision dosing.
In conclusion
So it is possible to extract psilocybin from mushrooms or truffles into a stable liquid in droplet form. Chemically, this requires the use of suitable solvents (preferably a water/alcohol mixture) and careful extraction methods that minimize degradation. Psilocybin is sufficiently polar to be highly soluble in aqueous media, and techniques such as cold maceration or ultrasonic extraction allow for high yields without destroying the compound. Once in solution, stability must be ensured: protect the extract from oxygen, light, and heat, maintain an alcohol percentage of at least ~20% for preservation, and optionally add antioxidants to protect psilocin. Under these conditions, psilocybin remains remarkably stable and effective in liquid form.
Dosage-based A droplet-form extract offers significant advantages: it allows the psychoactive substance to be measured with laboratory precision. Instead of relying on varying mushroom strengths, a standardized extract allows one to ingest exactly the desired number of milligrams of psilocybin, whether for microdosing or for a full psychedelic experience. Theoretically, the concentration can be adjusted so that a single drop produces a measurable effect, or multiple drops together reach the target value. This makes administering psilocybin just as dosable as, for example, a tincture or a medicinal droplet solution.
Note: The above description is intended as an educational exploration of possibilities and chemical principles, not as an encouragement or manual to carry out illegal extractions yourself. Psilocybin-containing mushrooms and truffles are classified as controlled substances in many countries. Making concentrated extracts entails additional risks and – if it is legal at all – should only be done in a safe, controlled environment with the proper knowledge. This text is limited to the theoretical framework and chemical feasibility.