Psilocybin, the active ingredient in magic mushrooms and truffles, is known as a psychedelic substance with deep consciousness-expanding effects. But psilocybin does much more than just induce a 'trip'. The substance also has powerful biochemical properties which can contribute to better physical and mental health — even in the absence of subjective experience.
In this article, we examine what psilocybin does at the cellular level, and what that could theoretically mean for the treatment or alleviation of various conditions.
1. Anti-inflammatory effects in the brain and body
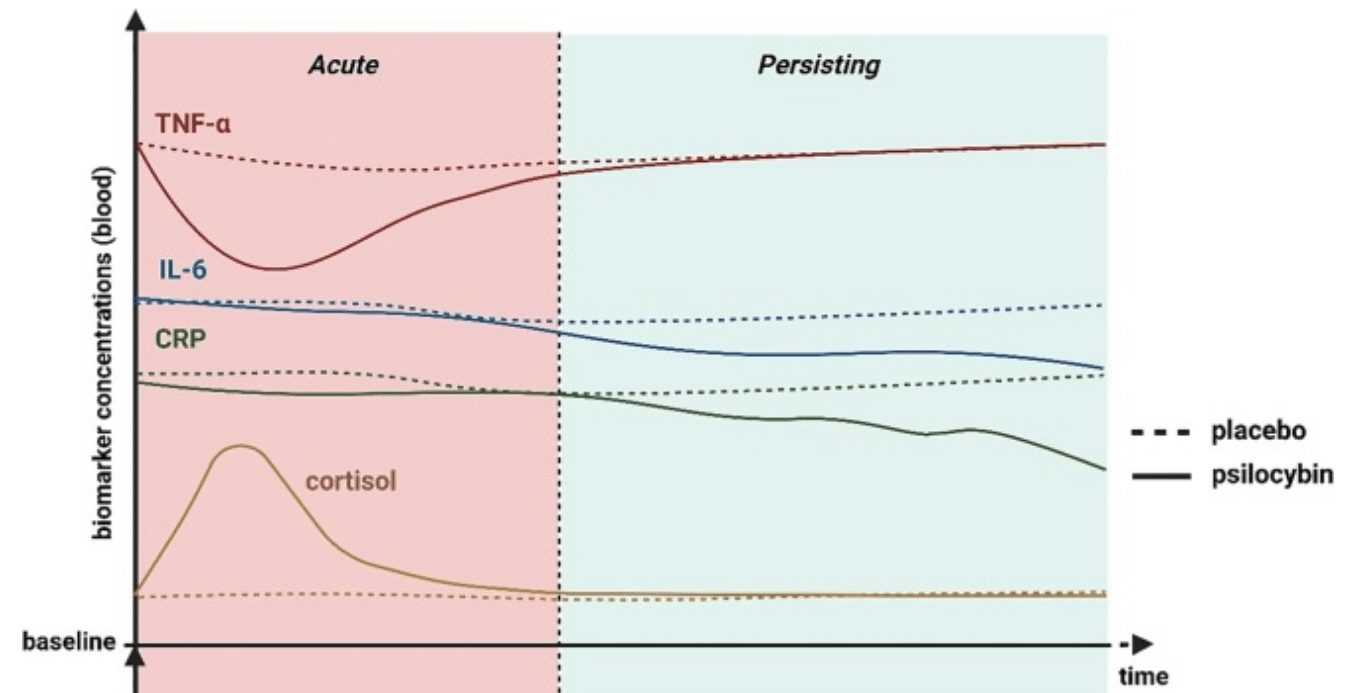
Recent research shows that psilocybin increases the levels of pro-inflammatory substances such as TNF-α, IL-6 and CRP lowers — substances that are often elevated in depression, anxiety, chronic stress, and neurodegenerative disorders. This anti-inflammatory effect is described in the article:
🔗 New mechanism of action of psilocybin discovered – anti-inflammatory in the brain
Biochemical mechanism:
-
Psilocin (the active substance after conversion of psilocybin) binds to 5-HT2A receptors on immune cells (including meningeal monocytes).
-
This inhibits inflammatory pathways via transcription factors such as NF-κB.
-
Result: reduction of both systemic and neuroinflammation.
Theoretically positive at:
-
Chronic inflammatory diseases (e.g. rheumatism, MS, Crohn's disease)
-
Depression and anxiety disorders (in which inflammation plays a role as an underlying mechanism)
-
Post-COVID symptoms with elevated inflammatory markers
-
Neurodegenerative diseases such as Alzheimer's and Parkinson's
See also the article by Tripforum about this link between inflammation and psychological complaints:
🔗 Psychedelics, inflammation, and the brain: new insights into anxiety and depression
2. Stimulation of neuroplasticity
Psilocybin increases the release of BDNF (Brain-Derived Neurotrophic Factor) and stimulates glutamate signaling, which leads to the formation of new synaptic connections in the brain — also known as neuroplasticity.
🔗 More about neuroplasticity and psychedelics
Mechanism:
-
5-HT2A activation → increased CREB → increased expression of BDNF and mTOR
-
This leads to the growth of dendrites, axonal branches, and synapses.
Possible health benefits:
-
Improved learning capacity, memory, and recovery after brain injury
-
Support for neurodegeneration (MS, Alzheimer's, Parkinson's)
-
Restoration of disrupted networks in depression, addiction, and trauma
3. Suppression of oxidative stress and cell aging
Psilocybin activates enzymes such as SIRT1, glutathione peroxidase (GPx) and superoxide dismutase (SOD), which together reduce oxidative stress and improve mitochondrial function.
What does SIRT1 do?
-
Repairs DNA damage
-
Extends telomere length
-
Reduces cellular senescence
Theoretically relevant to:
-
Aging processes
-
Conditions with high oxidative load (such as diabetes, cardiovascular disease)
-
Recovery after physical or mental burnout
-
Idiopathic pulmonary fibrosis (IPF)
4. Positive effects on the pancreas and type 2 diabetes
Although still experimental, there are indications that psilocybin the β-cells in the pancreas (which produce insulin) can protect against damage in type 2 diabetes.
🔗 What positive effects does psilocybin have on diabetes?
Important mechanisms:
-
Inhibition of TXNIP, a protein that damages β-cells at high glucose levels
-
Inhibition of apoptosis pathways via STATE1 and STATE3
-
Prevention of beta cell dedifferentiation (thereby allowing them to retain their function)
Implication:
-
Psilocybin could theoretically contribute to the maintenance of insulin production.
-
Potentially useful as adjunctive therapy for metabolic syndrome or prediabetes
5. Immunomodulation via serotonergic pathways
Immune cells such as T cells and microglia contain serotonin receptors (specifically 5-HT1A and 5-HT2A). Psilocybin influences the immune system via these receptors.
Effects:
-
Less overactivation of microglia (the brain's 'cleaner cells')
-
Balance between pro- and anti-inflammatory T-helper cells
-
Suppression of autoimmune reactions
Theoretically favorable with:
-
Autoimmune diseases (such as lupus, MS)
-
Neuroinflammation in chronic stress
-
Brain damage after trauma
Summary table: what can psilocybin mean biochemically?
| Mechanism of action | Biochemical route | Possible applications |
|---|---|---|
| Anti-inflammatory | ↓ TNF-α, IL-6, CRP via NF-κB inhibition | Anxiety, depression, post-COVID, MS |
| Neuroplasticity | ↑ BDNF, mTOR, glutamate | Depression, burnout, Parkinson's |
| Antioxidation & rejuvenation | ↑ SIRT1, ↓ oxidative stress | Aging, IPF, cell repair |
| Protection of β-cells | ↓ TXNIP, ↓ STAT1/3, preserve insulin function | Type 2 diabetes, metabolic syndrome |
| Immunomodulation | 5-HT2A on T-cells and microglia | Autoimmune diseases, encephalitis |
Conclusion: a molecular medicine in the making?
Although psilocybin is best known as a psychedelic substance, the scientific data point to broad biological effects on inflammation, neuroplasticity, oxidation, cell death, and immunity. These effects occur via known molecular pathways and are not limited to the psyche — the body itself also undergoes measurable changes.
The therapeutic value of psilocybin thus extends beyond spiritual experiences or psychological insights. As a regulator of cell health and immune balance psilocybin offers a promising angle for future medical applications, although large-scale clinical research remains necessary.
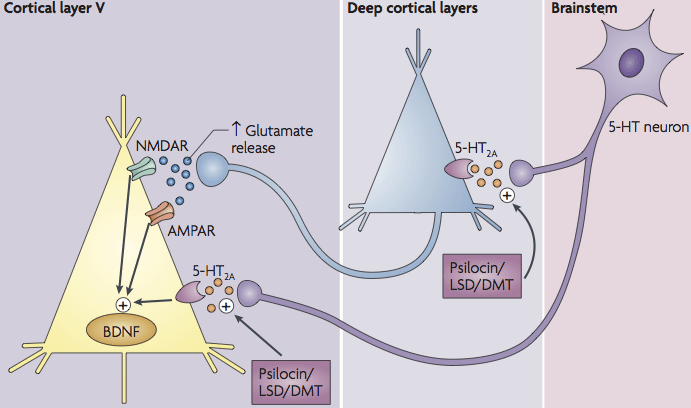
Serotonin 5-HT2A Receptor and Signal Transduction
Psilocybin (via its active metabolite psilocin) exerts its primary action as an agonist of the serotonin 5-HT2A receptor in the brain. Upon stimulation, this G-protein-coupled receptor (Gq) activates intracellular signaling pathways such as phospholipase C, leading to an increase in inositol triphosphate (IP₃) and calcium release in neurons. This cascade activates various kinases (e.g., PKC, MAP kinases), which can influence gene expression and promote neuronal plasticity. 5-HT2A receptors are abundantly present on cortical pyramidal cells (especially in layer V of the prefrontal cortex) as well as on GABA interneurons. Activation of 5-HT2A in these networks ultimately causes increased neuronal excitation – partly through direct stimulation of glutamatergic pyramidal cells, partly through disinhibition (inhibition of local GABA interneurons). The result is an increased cortical signal transmission and disruption of normal rhythms, which is associated with the psychedelic changes in consciousness. Classical research showed, for example, that serotonin via 5-HT2A The excitatory postsynaptic current in prefrontal pyramidal cells is increased by asynchronous glutamate release. This mechanism underlies the acute sensory and cognitive effects of psilocybin.
5-HT2A activation immediately upregulates certain genes (so-called immediate early genes) involved in plasticity. For example, it has been shown that psilocybin acutely affects the expression of EGR1 increases, a gene that facilitates neural growth and synapse formation. Furthermore, 5-HT2A plays a crucial role in downstream signal transduction to neuroplastic pathways: it triggers the release of neurotransmitters and second messengers that, for example, stimulate the production of neurotrophins. Importantly, the hallucinogenic effects are strictly 5-HT2A-dependent: in animal models, the typical head-twitch response (correlation of hallucinations) is blocked by 5-HT2A antagonists. Psilocybin also binds to a lesser extent to other serotonin receptors (such as 5-HT1A), which may contribute to anxiolysis and mood regulation, but the 5-HT2A pathway dominates the unique consciousness-expanding effect.
Glutamate and Neurotransmission
Glutamate is the principal excitatory neurotransmitter in the central nervous system, and psilocybin strongly influences glutamatergic transmission via 5-HT2A mechanisms. Activation of 5-HT2A on glutamatergic pyramidal cells leads to increased glutamate release in cortical circuits. This causes a cascade of excitation in neural networks and increases neural plasticity and information processing. Simultaneously, 5-HT2A activation on inhibitory interneurons can reduce inhibition, which a net increase in glutamatergic signaling brings about. Studies support this: classic psychedelics cause asynchronous, increased glutamate release in the prefrontal cortex. This acute glutamate surge is associated with the “desynchronized” brain state and increased cortical connectivity under psilocybin.
Interestingly, the glutamate effects of psilocybin appear to be region-specific and dynamic. A high-resolution MRS (magnetic resonance spectroscopy) study showed that immediately after administration the glutamate level in the hippocampus decreased, and that acute glutamate decline correlated with a reduction in TNF-α. This suggests that psilocybin possibly tempers excessive glutamate signals in stress-related memory centers (hippocampus), while elsewhere it actually increases signal transmission. Moreover, increased glutamate release, via the activation of AMPA receptors, can trigger downstream pathways that promote plasticity. For example: 5-HT2A-stimulated glutamate release activates AMPA receptors, which in turn the issuance of brain-derived neurotrophic factor (BDNF) triggers and stimulates TrkB receptor signaling. Thus, glutamate forms a crucial link between initial receptor activation and the broader cellular response, establishing the connection between neurotransmission and growth factor-driven repair mechanisms.
Neuroplasticity and Neurotrophins (BDNF, mTOR)
Neuroplasticity refers to the ability of brain cells to form new connections and change structurally/functionally. Psilocybin appears to be strong plasticity-promoting properties to have. For example, classic psychedelics (psilocybin, LSD, DMT, etc.) increase dendritic branching and dendritic spine density of neurons in the prefrontal cortex, comparable to or even stronger than ketamine (a well-known fast-acting antidepressant). In culture experiments and animal models, psychedelics stimulate the outgrowth of neurites and the formation of new synapses, giving them the name “psychoplastogens” have received. This effect is directly linked to certain molecular growth signals.
An important role is reserved for the neurotrophin BDNF (brain-derived neurotrophic factor) and its receptor TrkB. Psilocybin increases the expression and release of BDNF, which is essential for synaptic growth and neuronal survival. In mice, a single dose of psilocybin has been shown to completely counteract the stress-induced decline in hippocampal BDNF levels. As a result, structural abnormalities caused by fear conditioning (such as fewer dendritic branches and spines) were prevented. Mechanistic target of rapamycin (mTOR), a central kinase in cell growth and synapse formation, is also activated in the process: in the same study, psilocybin restored the decrease in mTOR protein in the hippocampus caused by traumatic stress. This indicates that psilocybin the BDNF-TrkB-mTOR route stimulates to promote neuroplasticity.
It is crucial that these plasticity effects depend on BDNF/TrkB and mTOR. It has been established experimentally that psychedelics lose their growth-stimulating effect if the TrkB receptor is blocked or if mTOR is inhibited. In other words, Psychedelically-induced synaptogenesis requires TrkB activation and mTOR signaling.. The 5-HT2A receptor also proves to be a necessary link in this process: blocking 5-HT2A prevents psychedelic-induced neurite growth and increase in spines. These three factors – 5-HT2A, BDNF-TrkB, and mTOR – thus function as a neuroplasticity node by psilocybin.
Remarkably, recent research suggests that psilocin (the active metabolite) possibly directly allosterically modulate TrkB receptors itself. In a study, psilocin bound with high affinity to TrkB, thereby enhancing BDNF signals. The resulting neuroplastic and antidepressant effects occurred independently of 5-HT2A, whereas the psychedelic sensory effects (such as hallucinations) remained 5-HT2A-dependent. This points to an interesting dualistic mechanismPsilocybin can induce acute changes in consciousness via the serotonergic pathway and support long-term neuronal recovery mechanisms via direct TrkB activation. Ultimately, these processes result in increased synaptic connectivity, improvement of emotional network circuits, and potentially long-lasting therapeutic effects (e.g., antidepressant results) after just one or a few doses.
Inflammatory Responses and Immunomodulation (TNF-α, IL-6, CRP, STAT1/3)
There is increasing evidence that psilocybin dampens inflammatory responses and influences immunological markers, which is relevant because many psychiatric disorders are associated with chronic inflammation. In a placebo-controlled study with healthy volunteers, a single dose of psilocybin was found to acutely lower the circulating levels of the pro-inflammatory cytokine TNF-α to significantly lower. Other inflammatory markers (such as IL-6, IL-1β and the acute-phase protein CRP) did not change immediately, but showed striking decreases after a week: seven days after psilocybin administration were IL-6 and CRP persistently lower compared to placebo. Interestingly, stronger decreases in IL-6 and CRP correlated with more persistent improvements in mood and social functioning of the participants. This suggests that the anti-inflammatory effects psilocybin may contribute to the long-term psychological benefits (such as antidepressant or anxiolytic effects) that are observed.
The immunomodulatory effect of psilocybin is supported by cellular studies. In human macrophage models (THP-1 cells) activated with LPS endotoxin, psilocybin suppresses the inflammatory cascade. In particular, it was observed that psilocybin inhibits the phosphorylation (activation) of important transcription factors such as NF-κB, STAT1 and STAT3 reduces. As a result, the gene expression of pro-inflammatory mediators decreased: psilocybin dose-dependently reduced the production of cytokines such as TNF-α and IL-1β in stimulated immune cells. This action occurs partly via inhibition of the IL-6/JAK2/STAT3 route, a central signaling pathway in inflammation regulation. By suppressing the TYK2/STAT3 pathway and simultaneously limiting STAT1 activation, psilocybin prevents the nuclear transcription of various inflammation genes. In summary, this indicates that psilocybin as an anti-inflammatory at the cellular level can work by blocking key pathways in cytokine production.
Also in the central nervous system Immune cells themselves are targets of psilocin. Microglia, the immune sentinels of the brain, also express 5-HT2 receptors. In microglia cell models, psilocin was found to act via these receptors. to temper reactive microglia. At non-toxic concentrations, psilocin had no effect on TNF-α secretion by activated microglia, but it did suppress phagocytic activity and significantly reduced the production of reactive oxygen and nitrogen radicals (ROS and NO). Two selective 5-HT2A agonists mimicked these effects, while 5-HT2A antagonists blocked them, confirming that these microglia modulation 5-HT2 driven is. This means that psilocybin is a neuroprotective component may have: by reducing microglial overactivation and oxidative stress, it can indirectly limit neuronal damage from inflammation. Overall, these findings support that psilocybin affects not only neurons but also glia and immune systems, resulting in reduced pro-inflammatory signals (TNF-α, IL-6, CRP) and possibly a more favorable environment for brain recovery.
Epigenetic Regulation and Gene Expression (SIRT1, TXNIP, DNA methylation)
In addition to acute receptor and signaling pathways, psychedelics can also long-term changes in gene expression and epigenetics bring about. There are indications that psilocybin (and related substances) modulate certain epigenetic mechanisms, although this field of research is still in its early stages. An example is the effect on DNA methylationLong-term therapeutic effects could be partially mediated by changes in DNA methylation patterns in brain cells. It has recently been reported that LSD, a similar classic hallucinogen, alters DNA methylation in neural tissues, resulting in altered expression of genes involved in neurotrophic and plasticity signaling pathways. Similar findings have been suggested for psilocybin. In a preclinical study in mice, it was found that the lasting social behavior-promoting effect of psilocybin could be blocked by a DNA methylation inhibitor, implying that epigenetic adaptations (such as methylation) were required to consolidate the effect. Although concrete data in humans are still lacking, there are ongoing epigenome-wide association studies to investigate whether psilocybin causes changes in methylation in blood or brain tissue. Such epigenetic changes could form a biological basis for the long-lasting resets in mood and cognition sometimes seen after psychedelic therapy.
Also at the level of histone protein modifications and enzyme regulation effects can occur. An important epigenetic and metabolic regulatory protein is SIRT1 (sirtuin-1), an NAD+-dependent histone deacetylase. SIRT1 is known for its anti-inflammatory and neuroprotective functions: it can deacetylate (inactivate) various transcription factors, including NF-κB and p53, thereby dampening oxidative stress and inflammatory responses. Although no direct research into psilocybin's effect on SIRT1 has been published to date, it is conceivable that activation of SIRT1 pathways plays a role in the observed anti-inflammatory effect and promotion of cellular health by psilocybin. Specifically, SIRT1 is activated under conditions of reduced stress and improved cellular metabolism, which could be associated with the normalization of stress markers after psychedelic use. Future research must determine whether psilocybin influences the expression or activity of sirtuins – for example, via the increase in NAD+ levels or via downstream signaling pathways – which would contribute to epigenetic modulation of neuronal functions.
Another factor of interest is TXNIP (thioredoxin-interacting protein), a protein that acts as a sensor coupling between cellular stress (such as high ROS or glucose levels) and inflammatory activation. TXNIP is a key switch that detaches from thioredoxin upon oxidative stress, thereby... NLRP3 inflammasome activates, which leads to cytokine production and cell inflammation. Chronic stress and depression are associated with elevated TXNIP levels and overactivation of inflammasomes. There are no publications yet that have explicitly measured the effect of psilocybin on TXNIP. However, given the aforementioned decrease in pro-inflammatory cytokines and ROS production under psilocybin, it is speculative that less TXNIP activation takes place (since TXNIP activation is often ROS-driven). In other words, by reducing oxidative stress and inflammation, psilocybin could indirectly inhibit the TXNIP-NLRP3 pathway, similar to what has been observed with other anti-inflammatory interventions. This remains hypothetical for the time being; future neuroimmunological studies (for example, in animal models of neuroinflammation) must determine whether psilocybin measurably alters TXNIP expression or inflammasome activity.
All in all, psilocybin also seems to be on the level of gene regulation to ensure shifts that promote recovery. Acute triggers (such as 5-HT2A activation, glutamate wave) activate or inhibit transcription factors, leading to changes in gene expression profiles in neurons and immune cells. Many of these genes code for neurotrophins (BDNF), growth factor receptors (TrkB), cytoskeletal proteins, or inflammatory mediators. This concert of changes – potentially secured by epigenetic markers (histone modifications, DNA methylation) – would be ”reprogramming” can support neural circuits that contribute to the long-lasting effects of psilocybin.
Rol of Psilocybin in the Central Nervous System
Psilocybin therefore intervenes in the neurobiology of the at multiple levels. central nervous system. Primarily, it acts as a potent serotonergic stimulator (via 5-HT2A) that causes acute changes in neurotransmission brings about – specifically increased glutamate activity in cortical networks – which explains the remarkable expansion of consciousness and sensory effects. At the same time, this receptor activation sets a chain of signal transduction set in motion that fuels cell growth and survival pathways. By stimulating BDNF production and TrkB receptor activation, and activating mTOR, psilocybin initiates a cascade that leads to synaptogenesis and dendritic growth. This results in increased connectivity and flexibility of neural networks – a phenomenon associated with the breaking of rigid negative thought patterns and the “resetting” of pathological brain functions in depression and other disorders.
Moreover, psilocybin exercises systemic and central anti-inflammatory effects out. By suppressing pro-inflammatory cytokines (such as TNF-α, IL-6) and modulating immune cells (microglia, macrophages), it creates an environment that promotes neurogenic repair processes. Chronic inflammation and stress hormone dysregulation (HPA axis hyperactivity) are known as promoting factors for neuronal atrophy and psychiatric symptomatology; psilocybin's normalizing influence on these may therefore be directly linked to its therapeutic potential.
Finally, the (preliminary) findings regarding epigenetics and gene expression suggest that psilocybin may permanent molecular changes triggers that put the brain in a more resilient state. It could “recalibrate” certain gene programs – for example, by activating neurotrophin genes and suppressing inflammatory genes – and prolong these changes via epigenetic markers (such as DNA methylation). Such mechanisms would explain how a single or limited number of doses of psilocybin long-term behavioral and mood changes can induce. Although many of these pathways still need to be investigated further, current evidence paints a picture of psilocybin as a pleiotropic substance that restructures neural circuits, restores neurochemical balance, and optimizes the cellular environment in the CNS. This unique combination of effects – on neuroplasticity, neurotransmission, immunomodulation, and possibly an epigenetic “reset” – forms the neurobiological basis for the promising results seen with psilocybin interventions in clinical research.
Conclusion: Psilocybin acts on various molecular pathways in the brain. Via 5-HT2A coupled signal transduction It directly influences neurotransmitter release (especially glutamate) and activates cellular growth processes. It increases neurotrophins such as BDNF and activates it mTOR-pathway, which leads to synaptic strengthening and neuroplasticity. In addition modulates the immune system by inflammatory markers such as TNF-α, IL-6 and CRP to lower and transcription factors as STAT3/STAT1 and to suppress NF-κB. It may also contribute to epigenetic reprogramming of neuronal gene expression, including via pathways involving enzymes such as SIRT1 and mediators as TXNIP are involved (although this has not yet been definitively demonstrated). Together, these mechanisms support the neuroplasticity, neurotransmission and anti-inflammatory in the central nervous system, giving psilocybin a unique therapeutic profile with potentially lasting positive effects on brain function and psychological well-being.
References
- Mason NL et al. (2023) – Brain Behavior Immune, research into psilocybin's effects on immune markers (TNF-α, IL-6, CRP) in healthy volunteers.
- Du Y et al. (2023) – Chin Med J, mouse study demonstrating that psilocybin promotes neuroplasticity via BDNF and mTOR signaling pathways and restores stress-induced neurodescent.
- Ly C et al. (2018) – Cell Reports, preclinical evidence that psychedelics (incl. psilocin) rapid structural plasticity induce; require 5-HT2A activation, TrkB (BDNF receptor) and mTOR.
- Castrén E et al. (2023) – Nature Neuroscience, discovery that psilocin directly to TrkB binds and strengthens BDNF signals; plasticity and antidepressant behavior due to psychedelics appeared to be dependent on TrkB/BDNF and independent of 5-HT2A.
- Wiens KR et al. (2024) – Molecules, study in microglia cells in which psilocin suppresses phagocytosis, ROS and NO production by activated microglia via 5-HT2A receptors (neuroinflammatory modulation).
- Ghasemi Gojani E et al. (2024) – Psychoactives (MDPI), in vitro inflammation model: psilocybin inhibits LPS-induced inflammation in macrophages via a decrease in phospho-NF-κB, STATE1 and STATE3, and associated decrease in cytokine expression.
- Raval NP et al. (2023) – Front. Immunol., review about SIRT1 in inflammation regulation; highlights SIRT1's anti-inflammatory action through deacetylation of histones and inflammatory factors.
- Shen X et al. (2018) – Nat. Rev. Cancer, describes TXNIP as a key molecule that links cellular stress to NLRP3 inflammasome activation. (Indirectly relevant to psilocybin's anti-inflammatory effect.)
- Dakic V et al. (2021) – Transl Psychiatry, showed epigenetic changes (DNA methylation, gene expression) after LSD treatment, with impact on neuroplasticity genes. (Related finding, possible analogue for psilocybin.)