Psychedelics and BDNF
Psychedelics such as magic mushrooms, truffles, LSD or psilocybin stand known to release more BDNF in the brain. As we described earlier, increased BDNF can have a beneficial effect on neurodegenerative disorders such as Parkinson's, Alzheimer's, and MS.
What is the interaction between the TAU protein and BDNF?
BDNF is known for its role in promoting the survival and growth of neurons, particularly dopaminergic neurons. These dopaminergic neurons are important because they are affected in Parkinson's disease. Therefore, in the context of Parkinson's disease, BDNF could influence and protect the survival and health of dopaminergic neurons.
Tau protein is usually found in elevated concentrations in other neurodegenerative disorders, such as Alzheimer's disease, in which Tau protein deposits are found in the brain. In Alzheimer's disease, there is evidence that Brain-Derived Neurotrophic Factor (BDNF) may influence Tau protein, albeit indirectly. Alzheimer's disease is characterized by the abnormal accumulation and aggregation of Tau protein and beta-amyloid protein in the brain. BDNF plays an important role in promoting the survival and health of neurons in the brain, and it may have various effects on Tau protein in the following ways:
-
Protection of neurons: BDNF can promote the survival and health of neurons, including those involved in Tau protein transport. This can help prevent the harmful effects of Tau protein accumulation.
-
Regulation of Tau phosphorylation: Tau protein must normally be correctly phosphorylated to perform its function in stabilizing microtubules in neurons. Abnormal phosphorylation of Tau can lead to the formation of Tau tangles, a hallmark of Alzheimer's disease. BDNF can influence Tau phosphorylation and potentially help prevent pathological changes in Tau protein.
-
Stimulation of neuroplasticity: BDNF can promote neuroplasticity, which is the brain's ability to learn and adapt to changes in the environment. This can indirectly reduce the severity of Alzheimer's symptoms.
More information about BDNF
Also read our latest post about BDNF or neuroplasticity via the following links:
Effects of psilocin on Alzheimer's and TAU proteins
Current scientific studies
Impact on neurodegenerative diseases (Alzheimer's):
Recent studies suggest that psilocybin (and its active metabolite psilocin) may have potentially beneficial effects on neurodegenerative disorders such as Alzheimer's disease (AD). Although direct research into psilocin in Alzheimer's While its use is still limited, reviews and preclinical studies show that classic psychedelics can stimulate neurogenesis, promote neuroplasticity, and reduce neuroinflammation. This is relevant because AD is characterized by synapse loss, neuronal death, and inflammatory processes in the brain. Psychedelics such as psilocybin are powerful agonists of serotonin (5-HT)2A receptors and induce broad changes in brain networks and plasticity. There is anecdotal evidence that microdosing (sub-perceptual doses) of psychedelics can improve cognition, which sparks interest for application in early dementia. In a mouse model of hippocampus-based learning For example, it has been found that both low and high doses of psilocybin accelerate the complete extinguishing of conditioned fear responses, indicating improved cognitive flexibility. Catlow et al. (2013) also observed that psilocybin stimulated the formation of new neurons in the hippocampus (hippocampal neurogenesis) in mice, which was associated with better memory retrieval. Such results support the idea that psilocybin/psilocin may be beneficial for neuronal health and memory functions.
Influence on TAU protein aggregation:
The direct effect of psilocin on TAU protein pathology is still not extensively investigated. AD is characterized by intracellular accumulations of hyperphosphorylated tau protein (neurofibrillary tangles) which, together with amyloid plaques, contribute to neuronal dysfunction. Specific studies demonstrating that psilocin reduces tau aggregation are scarce. However, there are indications from related research that psychedelics may indirectly influence tau pathology. preclinical example This comes from a rat model of mild brain trauma: treatment with psilocybin led to reduced levels of hyperphosphorylated tau in the brain. Furthermore, in that study, the concentration of BDNF (brain-derived neurotrophic factor) increased – a growth factor known to protect against tau-related neurodegeneration. Additionally, it is known that certain beta-carboline psychedelics (such as harmine, a component of ayahuasca) can reduce tau phosphorylation at AD-related sites. These findings suggest that activation of certain cellular pathways by psychedelics may counteract tau accumulation, although psilocin itself still needs to be investigated further in this regard. To date, the focus of studies primarily on symptoms and general neuroprotection, not directly on tau clearance; future research (for example, in animal models with tau pathology) is needed to confirm whether psilocin can reduce tau tangles.
Research methods (in vitro, in vivo, clinical):
Current knowledge comes from a mix of laboratory research and early clinical explorations. In vitroStudies have investigated the cellular effects of psilocin: for instance, a study using microglia (brain immune cells) showed that psilocin can modulate inflammatory markers. In cultured microglia, psilocin significantly reduced the production of reactive oxygen species (ROS), as well as the release of nitric oxide (NO), via activation of 5-HT2 receptors. These cell models support an anti-inflammatory effect (relevant because chronic neuroinflammation contributes to AD). In vivoStudies (animal models) show that psilocybin/psilocin can improve neurological functions. Experimental administrations have been used in mice and rats to measure effects on neuroplasticity and behavior. Examples include the aforementioned mouse studies in which psilocybin stimulated neurogenesis and accelerated learning processes, and rat studies in which psilocybin limited brain damage caused by trauma (fewer MRI abnormalities, less tau pathology). Also, in a rat model of streptozotocin-induced Alzheimer's (STZ-AD model), 5-HT1A/2A agonists proved neuroprotective: they prevented cell death in the hippocampus via anti-apoptotic and anti-inflammatory pathways. This suggests that agents such as psilocin, which act on the same receptors, may offer comparable protection. Clinical studies are still in their infancy. To date, there are no large-scale clinical trials demonstrating that psilocin effectively alters AD processes. However, there are ongoing small-scale clinical trials aimed at symptomatic improvement: for instance, a pilot study has been initiated to test psilocybin in patients with mild cognitive impairment (MCI) or early Alzheimer's who suffer from depression (NCT04123314). This study primarily investigates whether psilocybin safely reduces depressive symptoms and improves quality of life in early-stage dementia. Results are expected in late 2023. Additionally, there is interest in the application of microdosing in older adults with cognitive decline, but this is currently at an anecdotal and exploratory level. Table 1 below provides an overview of representative studies and their findings:
| Study (year) | Model / method | Key findings (relevant to AD/TAU) |
|---|---|---|
| Shahidi et al., 2019 | Rat model (STZ-induced Alzheimer's) | 5-HT1A/2A receptor agonists protected hippocampal neurons via anti-apoptotic and anti-inflammatory effects. |
| Catlow et al., 2013 | Mice (trace fear conditioning task) | Psilocybin increased hippocampal neurogenesis and accelerated the extinction of fear-conditioned memories. |
| Shao et al., 2021* | Mice (acute administration of psilocybin) | Psilocybin (1 mg/kg) increased the density of dendritic spines in the frontal cortex (effect stronger in females). |
| Wiens et al., 2023 | In vitro – microglial cells | Psilocin (≤10 µM) suppressed the respiratory burst in human microglia-like cells (reduced ROS production) and reduced NO release via 5-HT2 receptor activation. |
| Ferris et al., 2025 | Rat model, repeated mild brain trauma | Psilocybin (3 mg/kg, ip) administration reduced hyperphosphorylated tau (NFT markers) and increased BDNF levels; also improvement in MRI biomarkers after trauma. |
| NCT04123314 (ongoing) | Clinical – patients with MCI/early AD + depression | Psilocybin-assisted therapy to alleviate depression in early-stage Alzheimer's (focus on mood and quality of life). Status: Phase 1 safety study; results pending. |
Footnote: Shao et al. 2021 refers to recent work cited in the literature; exact reference year subject to change.
Theoretical mechanisms of action
Neuroprotective effects of psilocin:
Based on the above studies and known pharmacology, several mechanisms have been proposed by which psilocin may be neuroprotective. First psilocin promotes neuroplasticity. Under the influence of psilocybin/psilocin, signaling pathways in neurons are activated, leading to the growth of new connections and the strengthening of synapses. Animal studies show an increase in dendritic spines (protrusions on neurons that make synaptic contact) after psilocybin use. There is also an increase in neurotrophic factors such as Brain-Derived Neurotrophic Factor (BDNF) observed. BDNF supports neuronal survival and promotes repair; interestingly, higher BDNF levels correlate with protection against tau-induced neurodegeneration. Secondly, psilocin has anti-inflammatory and antioxidant properties. Excessive neuroinflammation is a driving force behind damage in Alzheimer's. Psilocin can inhibit this process via microglia modulation: exposure of microglial cells to psilocin reduces the production of pro-inflammatory molecules (such as ROS and NO) that would otherwise contribute to oxidative stress and cell damage. This indicates a neuroimmunomodulatory effect, which can benefit neuronal survival in an inflamed AD brain. Furthermore, models suggest that 5-HT_2A agonists trigger anti-apoptotic signals, causing neurons to undergo programmed cell death less rapidly in disease models. Thirdly, psilocin can indirectly cognitive functions improve by alleviating symptoms such as depression and anxiety. AD patients with less depression/anxiety show slower cognitive decline; psilocybin is known to provide strong and lasting improvements in depressive symptoms in treatment-resistant patients in other contexts. By improving mood and anxiety, psilocin could also have a secondary protective effect on cognitive decline.
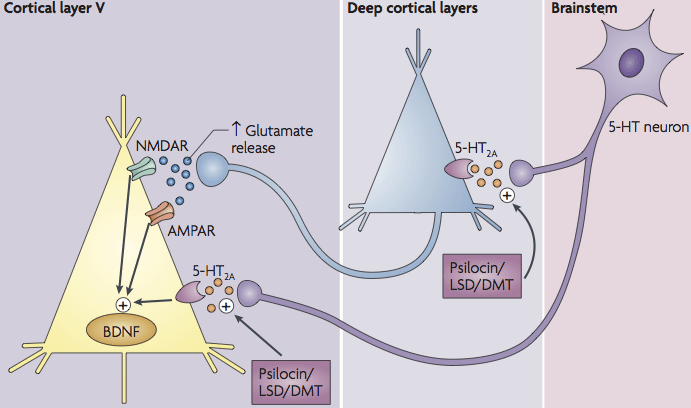
Biochemical pathways (TAU protein, neuronal health):
Several cellular pathways may be involved in the effects of psilocin, which may be relevant to tau protein accumulation and neuronal health. An important pathway is the BDNF/TrkB signal coupled to mTOR. When psilocin increases neuronal activity via 5-HT-2A receptors, this can lead to glutamate release and activation of TrkB (the BDNF receptor). This activates the mTOR pathway, resulting in phosphorylation of CREB (a transcription factor) and increased expression of genes associated with synaptic growth and plasticity. This mechanism partially explains the psychoplastogenic effects (structural brain plasticity) of psychedelics. However, mTOR plays a complex role in Alzheimer's: excessive mTOR activity can inhibit autophagy, a process necessary to clear misfolded proteins. In AD patients, hyperactive mTOR has been observed to be associated with increased amyloid-β and tau phosphorylation. Theoretically, therefore, it is important that treatment with psilocin well the plasticity increases but not pathologically overactivates mTOR. As yet, there is no evidence that psilocybin has these types of adverse effects – it is even speculated that psilocybin does so via controlled mTOR activation could actually facilitate the removal of protein aggregates. In a 5xFAD mouse model (familial AD), for example, activation of mTOR in microglia was shown to increase TREM2 expression and promote amyloid plaque clearance. Psilocin might potentially utilize a similar mechanism to support amyloid and tau degradation, but this remains hypothetical for now. Other biochemical pathways: Serotonergic activation via 5-HT_2A can also influence gene expression patterns that reduce neuronal stress. For example, stimulation of 5-HT_2A receptors can the SIRT1/PGC-1αactivate the pathway in neurons, which improves mitochondrial biogenesis. Better mitochondrial function means less oxidative stress and potentially less neuronal damage, which can indirectly limit the accumulation of tau (since stress kinases hyperphosphorylate tau). Additionally, there are indications that serotonergic modulation can influence enzymes that phosphorylate tau. Although not specifically studied with psilocin, chronic inflammation and stress kinases (such as GSK-3β, CDK5) are known to contribute to tau pathology; the anti-inflammatory effect of psilocin may break this cascade. In summary, the biochemical pathways suggest that psilocin is multi-factorial effect may have: it promotes pro-survival and pro-plasticity signals (BDNF/CREB, SIRT1), while dampening harmful processes (neuroinflammation, oxidative stress, possibly tau phosphorylation via intermediate steps).
Roll of 5-HT2A-receptor activation in neurodegeneration:
The 5-HT_2A receptor is the primary target site of psilocin/psilocybin and plays a key role in the mechanisms discussed. Interestingly, post-mortem and PET studies have shown that the density of 5-HT_2A receptors is reduced in Alzheimer's patients, and this decrease correlates with cognitive decline. This suggests that serotonergic signaling – particularly via 5-HT_2A – is involved in cognitive function and possibly in the pathogenesis of AD. Activation of 5-HT_2A receptors has a wide range of effects: in cortical neurons, it can lead to the release of glutamate, modulation of gene expression (including immediate early genes such as c-Fos), and stimulating growth pathways (as described above). In addition, 5-HT_2A often forms receptor complexes with other receptors (e.g., mGlu2 or 5-HT1A), which exerts subtle influences on signaling pathways. For neurodegeneration, this means that 5-HT_2A agonism potentially both symptomatic lighting as disease modification can offer. Symptomatic, because 5-HT_2A activation in the frontal cortex and hippocampus (areas affected in AD) can temporarily strengthen networks and, for example, reduce apathy or depressive symptoms. Disease-modifying, because neuronal survival mechanisms are triggered via 5-HT_2A (such as BDNF production) and immune responses in the brain can be shaped. An example is the effect on microglia: psilocin's inhibition of microglia-ROS production was mimicked by selective 5-HT_2A agonists, confirming that precisely this receptor pathway is responsible for the anti-inflammatory effect. The therapeutic benefit The role of 5-HT_2A activation would therefore lie in restoring neurochemical balance that is disrupted in AD. Nevertheless, the hallucinogenic effects of psilocybin are a direct consequence of 5-HT_2A agonism in cortical circuits, which is challenging in vulnerable patients. Researchers are therefore investigating whether 5-HT_2A stimulation with moderate intensity (for example via microdosing or via new compounds that activate the receptor complex differently) is sufficient to achieve neuroprotective benefits without strong sensory disturbance. In summary, 5-HT_2A plays a central role in the theoretical mechanisms whereby psilocin can promote neuronal health, and at the same time constitutes the source of potentially unwanted psychedelic side effects that must be managed.
Possible therapeutic applications
Research into psilocin as a treatment for Alzheimer's:
Based on the above, psilocin/psilocybin is viewed as a promising, albeit experimental, approach for AD. Preclinical data indicate multimodal benefits – from improved synaptic connectivity to reduced neuroinflammation – which are highly relevant for a disease like Alzheimer's, which is multifactorial. In theory, psilocin could be used to both the nuclear pathology to address (by promoting cleanup processes and protecting neurons) if the neuropsychiatric symptoms to alleviate AD (such as depression, anxiety, apathy). This makes it an attractive candidate for further study. However, current therapeutic applications are still primarily found in the research phase. As mentioned, there is an ongoing clinical trial using psilocybin in people with mild cognitive complaints and depression to see if this improves their well-being. In addition, there are small proof-of-concept Studies have been proposed to measure the effect on cognitive symptoms, for example, whether microdosing psilocin can stabilize memory function in early AD. To date, there is no officially approved use of psilocin for neurodegenerative diseases. The future application could look like an integrated approach: a combination of psilocybin-assisted psychotherapy to improve mood and neuroplasticity, possibly in microdosing or a modified form to limit the hallucinations. There are also initiatives to develop chemical analogs of psilocin that retain the therapeutic effects but are less psychedelic. This would be particularly useful for elderly people with Alzheimer's, where a full psychedelic experience might be burdensome or confusing. Despite the promising preclinical results, more research Required: long-term animal studies to assess effects on amyloid and tau patterns in the brain, as well as clinical studies in patients (initially focused on safety and proof-of-concept). Only then can it be assessed whether psilocin can find a place as an (adjunctive) therapy for Alzheimer's.
Side effects and risks: Psilocin (as a psychedelic) has known side effects, which must be weighed with extra care in the context of neurodegenerative patients. In general, psilocybin is in clinical trials well tolerated proven, with predominantly mild and transient side effects. Frequently reported are physiological effects such as increased blood pressure and heart rate, pupil dilation, dizziness, and sometimes headache after ingestion. During the psychedelic experience itself, psychological side effects occur, including anxiety, confusion, or an unpleasant “trip” if the setting is not optimal. In Alzheimer’s patients, who may already be confused or anxious, the risk of acute disorientation and panic is therefore a major concern. Rigorous medical supervision and a safe environment are necessary for any clinical use. In addition to these short-term effects, there are also some rare but serious risks reported (primarily with recreational or unsupervised use of high doses). Examples of this are cases of takotsubo cardiomyopathy (acute myocardial weakness) after use of hallucinogenic mushrooms, severe rhabdomyolysis (muscle damage) associated with psychedelics, the triggering of manic episodes in individuals with a predisposition to bipolar disorder, and occasionally long-term perceptual disturbances (HPPD: hallucinogen persisting perception disorder). Although these complications are rare, they emphasize that psilocin is not entirely risk-free, certainly not in a vulnerable population. Particular caution applies to neurodegenerative patients: comorbid health problems (such as cardiovascular conditions) can reduce tolerance to blood pressure spikes, and polypharmacy (multiple medications) can lead to unknown interactions with psilocin. Furthermore, clinical trials usually exclude people with a history of psychosis, because serotonergic hallucinogens could exacerbate a latent psychotic picture. Nevertheless, most controlled studies to date show that, provided patients are properly screened and monitored, serious side effects very unusual are associated with psilocybin use. In the context of Alzheimer's, the focus would likely be on lower dosages or non-hallucinogenic variants to minimize risks. Current studies are also paying attention to methods to attenuate the hallucinogenic component without losing the therapeutic effect. In summary, the main side effects of psilocin: acute psychedelic perceptual disturbances (including potentially frightening experiences), physiological stress response (increased heart rate/blood pressure), and, more rarely, severe reactions with unsupervised use. However, these risks can be mitigated by thorough screening, medical supervision, proper dosing, and set/setting—factors that are central in research settings.
Conclusion:
Psilocin and psilocybin constitute a new area of research in the context of Alzheimer's. Current scientific literature shows cautious optimism that these substances possess neuroprotective properties—ranging from stimulating synaptic growth to suppressing harmful neuroinflammation—that are relevant to counteracting AD pathology. A direct impact on tau protein aggregation has not yet been convincingly demonstrated, but indirect pathways (such as increased BDNF, reduced inflammation) may exert a positive influence on tau pathology. Theoretical mechanisms center around 5-HT_2A receptor activation and the cascade of cellular effects resulting therefrom, including gene expression changes favoring neuronal survival. Potential future therapeutic applications include both treating neuropsychiatric symptoms in dementia and inhibiting neurodegeneration itself, although hard clinical evidence for this is still lacking. Crucially, in the event of application, a balance must be struck between effects and side effects – for example, through innovations such as microdosing or new analogues – so that patients with Alzheimer's can benefit from the neuroprotective effects without disproportionate risks. Further preclinical and clinical studies in the coming years will have to determine to what extent psilocin can play a safe and effective role in the fight against Alzheimer's.
Sources: This overview is based on a synthesis of recent reviews and studies, including Frontiers in Neuroscience (2024), Frontiers in Synaptic Neuroscience (2020), Journal of Neurochemistry (2022), and experimental research published in, among others,. Experimental Brain Research, Molecules, and Molecular Psychiatry. The table of research results refers to specific studies and trial registries (see references in the table). These sources collectively endorse the potential of psilocin/psilocybin as an innovative approach for Alzheimer's, while simultaneously emphasizing that further research is needed to validate the preliminary findings.