Does psilocybin make your DNA younger?
Yes, there are strong indications that psilocybin can make your DNA “younger” at a cellular level, according to the article. psilocybin and telomere length. That article discusses in detail how psilocybin – or rather, the active ingredient psilocin – extends cell life and protects against the typical signs of aging, such as telomere shortening and oxidative damage.
Telomeres are the protective ends of your DNA strands. Every time a cell divides, these become slightly shorter, and when they become too short, the cell stops dividing or dies. Psilocybin appears to be able to slow down this wear and tear process. In a 2025 study, telomere length remained virtually stable in cells treated with psilocin, while it shortened significantly in the control group. At the same time, psilocybin increased the activity of the enzyme SIRT1, which stimulates DNA repair, activates telomerase, and suppresses inflammation, among other things. Especially in combination with a healthy lifestyle, a psilocybin session contribute to cell rejuvenation and slowing down biological aging.
If you want to use psilocybin with rejuvenation as the goal, Triptherapie advises doing a preliminary intake to do. This way you get personalized advice, including possible additions such as fisetine or NAD+ boosters that can enhance the effect on DNA and cell health.
See also:
Neural health and neurogenesis
Research suggests that psilocybin may have positive effects on neural health by promoting neuroplasticity and possibly neurogenesis. In a mouse model of post-traumatic stress (fear conditioning), a single dose of psilocybin (2.5 mg/kg) restored the growth of new nerve cells in the hippocampus: the number of young neuron markers (DCX+, BrdU+) increased compared to untreated animals. Additionally, psilocybin reduced the stress response in these mice (less “freezing” behavior) and restored reduced levels of BDNF (brain-derived neurotrophic factor) and mTOR to normal. This suggests that psilocybin can promote neuronal growth and repair under stress conditions, which directly points to neurogenic effects (in animals).
However, the dose proves to be crucial. An earlier study in mice observed a small increase in new neurons at a very low dose (0.1 mg/kg), but higher doses (0.5–1 mg/kg) lowered specifically the survival of new brain cells. This negative effect at high doses was blocked by a serotonin receptor antagonist (ketanserin), indicating that the action occurs via 5-HT receptors. The neurogenesis-promoting effects of psilocybin are therefore direct demonstrated in animal studies, but dependent on the dose and context. In humans, direct hippocampal neurogenesis is difficult to measure; however, clinical findings indirectly suggest that psilocybin makes the brain “more flexible.” For example, in patients with depression, a increase in cognitive flexibility and neural connectivity measured, indicating improved brain function and plasticity. These are indirect signs of “rejuvenation” of neural networks, although direct evidence for human neurogenesis is still limited.
Cellular aging (senescence, oxidative stress, mitochondria)
Recent laboratory studies provide first direct indications that psilocybin can slow down cellular aging. In an in vitro model of cellular aging (human fibroblasts), continuous exposure to psilocybin was shown to slow down aging. slow down: markers of cell senescence decreased and cell division capacity increased significantly. Specifically, psilocybin reduced the expression of cell cycle inhibitors (p16^INK4a^, p21) and the secretion of inflammatory factors by senescent cells (the so-called SASP, senescence-associated secretory phenotype). At the same time, the activity of DNA repair and proliferation markers (such as PCNA) increased. Also, less oxidative stress measured in treated cells, indicating better mitochondrial function and less accumulation of harmful reactive oxygen species. These results are direct (in cells) and suggest that psilocybin is anti-aginghas an effect at the cellular level. Importantly, this concerns a single cell line study; additional experiments (e.g., in different cell types or tissues) are needed to confirm the breadth of this effect.
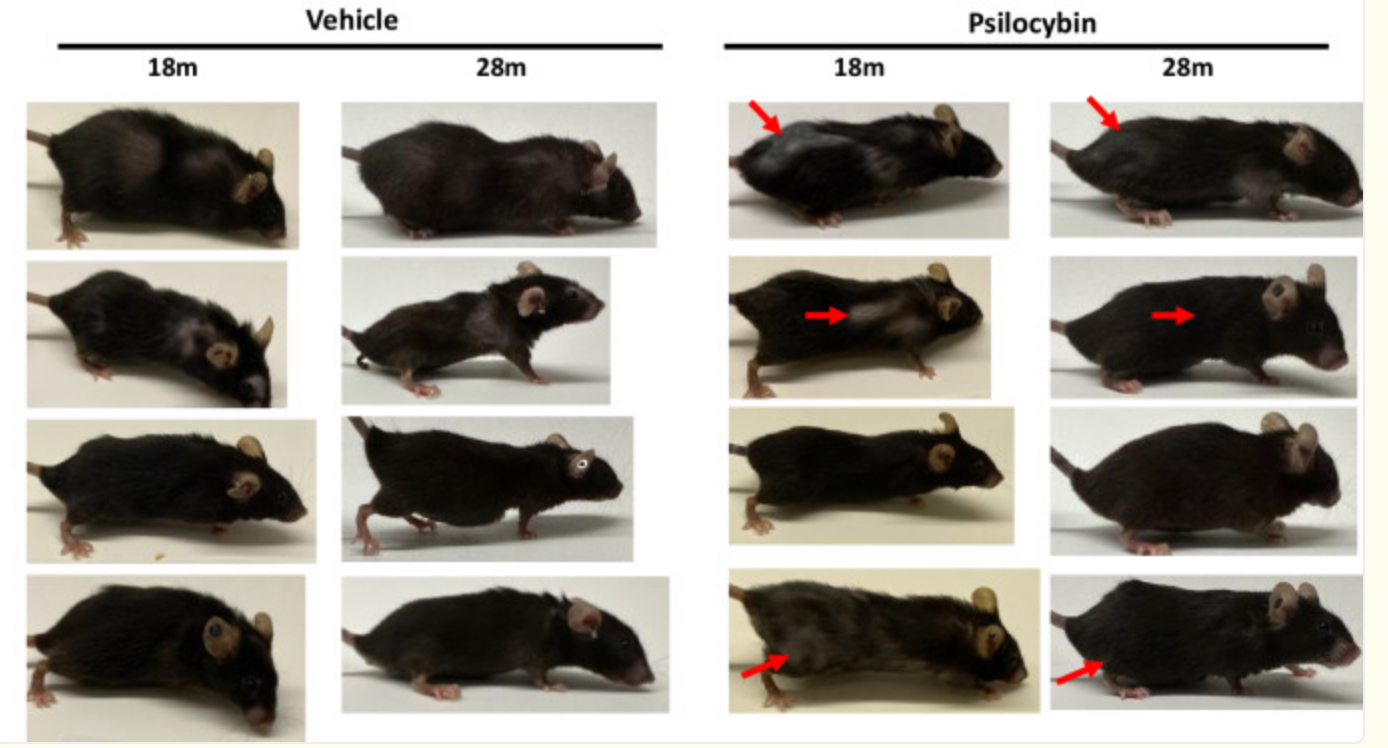
In addition, there is carefully evidence from animal research. In a recent study with older mice (approximately 60 “human years” old), psilocybin treatment prolonged the lifespan and improved physical signs of aging. Mice that received psilocybin monthly for 10 months had a survival rate of 80% compared to 50% in the control group, and remarkably, bald patches grew back and graying fur patches regained their pigment. These rejuvenation of the fur points to cell repair in skin/hair. Although this is impressive, it should be noted that the doses administered to mice were relatively high and the translation to humans is uncertain. In summary, the evidence for a cell-rejuvenating effect of psilocybin is encouraging but mainly limited to cell cultures and animal models; human confirmation is still lacking.
Telomere length and telomerase activity
Telomeres, the protective DNA end pieces of chromosomes, shorten with every cell division and serve as a molecular “clock” for aging. There is a hypothesis that psilocybin can slow down this clock by counteracting telomere shortening. These “psilocybin telomere hypothesis” This is supported by the fact that improved mental health often goes hand in hand with longer telomeres, whereas chronic stress and depression are associated with accelerated telomere shortening. Psilocybin can have strong positive effects on mental health (such as reducing anxiety and depression), so it was hypothesized that it could indirectly also preserve or lengthen telomere length.
Recent lab research offers first direct Evidence of influence on telomeres. In the previously mentioned cell study, human lung and skin fibroblasts that aged under the influence of psilocybin were found to, no significant telomere shortening to show compared to untreated senescent cells. In other words: psilocybin-treated cells retained telomere lengths comparable to young control cells, whereas in normal senescent cells the telomeres were clearly shortened. This indicates that psilocybin the telomere integrity can protect. Regarding the role of telomerase (the enzyme that lengthens telomeres) nothing has been directly reported in this context yet, but the preservation of telomere length suggests that psilocybin either increases telomerase activity or decreases the rate of telomere breakdown. Given the early stage of this research, the evidence here is limited for the time being. mediocreWe have a convincing cell model result, but there are no clinical studies yet that have measured telomere length in psilocybin users. Future research (for example, telomere measurements in participants of psilocybin therapy studies) is needed to test this promise in humans.
Inflammation and immune response
Psilocybin appears to have effects not only in the brain but also in the immune system – with potential anti-inflammatory results. In a placebo-controlled study with healthy volunteers, a single moderate dose of psilocybin acutely lowered the level of the pro-inflammatory cytokine TNF-α (tumor necrosis factor alpha) in the blood. Although other inflammatory markers (such as IL-1β, IL-6 and C-reactive protein) had not changed immediately after ingestion, there was a striking effect seven days later: the levels of IL-6 and CRP were significantly decreased compared to the placebo group. This indicates a lasting immunomodulatory mechanism of action of psilocybin. Interestingly, stronger decreases in IL-6 and CRP were associated with better mood and social functioning in the subjects one week after the session. These human data (n=30 psilocybin vs 30 placebo) provide direct evidence that psilocybin affects the immune system makes positive adjustments, albeit from a relatively small study.
Preclinical experiments confirm this picture of anti-inflammatory. In a 3D tissue model and immune cell experiments, it was found that psilocybin and its metabolite psilocin suppress the release of multiple pro-inflammatory mediators. For example, psilocybin significantly reduced the production of TNF-α and IFN-γ (interferon-gamma), as well as cytokines IL-6 and IL-8, which are involved in inflammatory responses. Furthermore, there are indications that psilocin beneficially influences microglia (the immune cells of the brain): it can inhibit an overactive, inflammation-promoting microglia status and even prevent microglia from clearing (phagocytosing) healthy neurons in an inflammatory model. All this points to the fact that psilocybin indirectly anti-agingcould have effects through the reduction of chronic inflammation, since low-grade inflammation (“inflammaging”) is a known factor in age-related diseases. The strength of evidence here is moderate to strongWe have a controlled human study for acute effects and multiple supporting lab studies. The mechanism appears to operate (partly) via serotonin receptors on immune cells that dampen inflammatory pathways. Further clinical research (for example, in patients with inflammatory conditions) will have to determine whether psilocybin can be used therapeutically to inhibit inflammation and associated aging processes.
Regulation of SIRT1 and other aging genes/proteins
A striking finding is that psilocybin influences genes and proteins associated with lifespan regulation. In cellular aging models, psilocybin led to increased expression of the enzyme SIRT1 (sirtuin-1), a protein known as a promoter of longevity and cell health. SIRT1 plays a key role in DNA repair, metabolism, and cellular stress response. In psilocybin-treated cells, SIRT1 levels increased significantly, whereas in aging control cells, SIRT1 remained low. Simultaneously, a decrease was observed. GADD45a, a gene that is activated by DNA damage, suggesting that less DNA damage or better repair occurred in the presence of or as a result of psilocybin. The amount also increased Nox4 (an enzyme that produces reactive oxygen) off, while No. 2 (a transcription factor that activates antioxidative genes) increased. These changes form a pattern: psilocybin appears cellularly the pro-survival to turn on a program that is also activated by caloric restriction or by substances like resveratrol (which also activate sirtuins). The activating SIRT1 and increasing Nrf2 indicates a immediate effect on epigenetic and stress response pathways that determine aging.
Besides SIRT1, there may be other related targets. For example, some researchers suggest that psilocybin via the HPA axis (stress axis) and glucocorticoid receptors influence gene expression, similar to what is seen during prolonged meditation. Although this is still hypothetical, it supports the idea that psilocybin intervenes broadly in aging pathways. Because the hard data currently largely come from single-cell studies, we consider the strength of evidence limited but intriguing. That research provides direct evidence that psilocybin favorably influences cellular “longevity genes,” but confirmation in vivo (in animals or humans) is still lacking. Nevertheless, this is a promising mechanism: activation of SIRT1 and related pathways may explain the diverse health benefits seen with psilocybin therapy. Further study is needed to determine whether this gene regulation also occurs at dosages and administration schedules used in a clinical context.
BDNF (Brain-Derived Neurotrophic Factor)
BDNF is a growth factor in the brain that is crucial for neuron survival, growth, and synaptic plasticity. There is growing evidence that psilocybin the BDNF signaling pathways activates and strengthens, which contributes to both antidepressant and potential anti-aging effects in the brain. In animal studies, for example, it has been shown that psilocybin can reverse the negative impact of chronic stress on BDNF: in anxious mice, a single dose of psilocybin restored stress-reduced BDNF expression in the hippocampus. This was accompanied by behavioral recovery (less depressive/anhedonic behavior) and increased neuronal branching, underscoring the role of BDNF in psilocybin's effects.
At the cellular level, psilocybin works via the 5-HT2A serotonin receptor, which activates a cascade that BDNF release and action promotes. In vitro research with nerve cells shows that 5-HT2A agonists (such as psilocybin/psilocin) the TrkB receptor, to which BDNF binds, activate and thus initiate growth processes. A remarkable recent discovery is that psilocin itself can bind directly to the TrkB receptor as a positive allosteric modulator. This means that psilocin influences the BDNF receptor in such a way that BDNF itself can bind and signal more effectively. Moliner et al. (2023) demonstrated that psilocin enhances the downstream effects of BDNF via this route, leading to increased dendrite growth and synapse formation. The result is improved neuronal survival and connectivity. Indirect evidence in mice supports this: LSD, a related psychedelic, increases the long-term survival of new hippocampal neurons via TrkB activation—a mechanism that has also been suggested for psilocin.
In short, psilocybin increases BDNFlevels and function in the brain, which can contribute to neuronal repair and cognitive function. This effect is likely crucial for therapeutic impact (such as antidepressant) and possibly for neuroprotection against aging. The strength of evidence here is reasonably strong in a preclinical sense: multiple animal studies and cell models confirm the involvement of BDNF. In humans, directly measuring BDNF in the brain is difficult, but a few studies have reported increased BDNF in the blood or improved cognitive flexibility (in which BDNF plays a role) after psilocybin. The indirect Evidence in humans is therefore in line with the preclinical findings. All in all, BDNF regulation appears to be important mechanism whereby psilocybin contributes to brain rejuvenation and recovery.
Glutamate regulation and synaptic plasticity
Glutamate is the principal excitatory neurotransmitter in the brain and plays a major role in synaptic plasticity (the ability of synapses to strengthen or weaken). Psychedelics, including psilocybin, influence glutamate metabolism., which leads to structural and functional changes in neural networks. For example, it is known that activation of 5-HT2A receptors on depth neurons can stimulate glutamate release in the prefrontal cortex, which triggers a cascade of synapse formation. This mechanism is similar to that of ketamine (another substance that promotes plasticity), albeit via a different receptor. The net effect is the activation of synaptic plasticity: new connections are formed between nerve cells and existing synapses are strengthened.
Strong evidence for this synapse-renewing effect comes from animal studies. In an experiment with pigs (a model that is closer to humans in terms of brain structure), a single administration of psilocybin measurably increased the number of synapses in the brain. One day after injection, the density of synaptic vesicle protein 2A (SV2A) – a marker for synapses – in the hippocampus ~4,4% higher than in control, and after one week the synapse density was increased even further (+9.2% in hippocampus, +6.1% in prefrontal cortex). This increase in SV2A binding indicates newly formed synapses which remained persistently present, reflecting a form of structural “rejuvenation” of the brain. Incidentally, it simultaneously appeared that psilocybin acutely reduced the density of 5-HT2A receptors (downregulation) – a likely feedback mechanism – but after a week that receptor adaptation had normalized again. The sustained synaptic gain was, however, striking and supports the idea that psilocybin can bring the brain into a more youthful, plastic state.
In addition, there are functional instructions for glutamate regulation by psilocybin in humans. A 7-Tesla MRS (magnetic resonance spectroscopy) study showed that glutamate concentration in the hippocampus decreased immediately after psilocybin intake. Interestingly, this acute glutamate drop correlated with the degree of TNF-α decline in the blood, suggesting that reduced neuronal excitation went hand in hand with a calming of immune activity. After a week, glutamate levels normalized again. In another study, a reduction in glutamate was measured in the anterior cingulate cortex (ACC) of depressed patients one week after psilocybin therapy, along with increased functional connectivity between brain networks. This indicates that psilocybin is a reset results in: first an acute disruption (desynchronization) of fixed networks and glutamatergic signals, followed by a reintegration with new synaptic connections. The net outcome is a more flexible neural network – something that could be considered a more “youthful” brain profile, because synaptic connectivity and brain dynamics are higher in young people.
The strength of evidence for glutamate-mediated synaptic plasticity psilocybin activity is high in a preclinical context (clear neuronal structure changes in animals) and reasonable in humans (neurochemical and connectivity changes measured, albeit in small test groups). These results strongly support the idea that psilocybin direct contributes to brain plasticity, which is essential for both functional recovery and the potential slowing of age-related cognitive decline.
Behavioral changes and cognitive/psychological rejuvenation
In addition to cellular and molecular effects, we also see with psilocybin behavioral and cognitive changes which suggest a rejuvenation of the mind. A characteristic of aging (or long-standing depression) is often cognitive rigidity: less flexible thinking and adapting. Studies show that psilocybin can counteract this. In an open-label clinical trial with depressed patients, psilocybin-assisted therapy improved the cognitive flexibility significant – measured as fewer perseveration errors in a set-shifting task – and this effect persisted for at least 4 weeks. This improvement occurred alongside mood improvement, but was not correlated one-to-one with it (suggesting that it is a separate positive effect). The increased ability to switch thought patterns is comparable to that of younger, healthy individuals, which can be interpreted as a psychologically rejuvenating effect. Similar observations have also been made in healthy individuals: after a single high dose of psilocybin, subjects reported increased openness of thought and creativity for months afterward, which is unusual because personality is normally stable after young adulthood. These types of changes—greater openness, cognitive fluidity, curiosity—are traits more commonly seen in younger people, and which often decline with aging or chronic stress.
Epidemiological research provides additional, albeit indirect, evidence. Data from a large cohort study in the US (MIDUS) showed that older adults who have ever used psychedelics on average performed better on executive functions and had fewer depressive symptoms than their peers who never used psychedelics. However, there was no difference in pure memory tests (episodic memory). Although this is observational and causal conclusions are difficult to draw (perhaps healthier, more innovative people were more inclined to try psychedelics), the picture aligns with clinical findings: psychedelic use correlates with better preservation of cognitive functions at a later age.
Regarding psychological well-being: psilocybin therapy is often accompanied by a strong and lasting reduction of negative mood states such as depression, anxiety, and existential distress (for example, in palliative care). In trials with cancer patients, researchers observed that a single psilocybin session resulted in drastically less anxiety and a higher sense of quality of life even after 6 months, something that could be described as restoring youthful zest for life and mental resilience. Moreover, ~70% of the participants consider their psilocybin experience to be one of the most personally significant events in their lives (comparable to the birth of a child). Such transformative experiences are often accompanied by positive behavioral changes – greater social connectedness, life purpose, and optimism – reminiscent of a “reset” of ingrained, age-related negative thought patterns.
Evidential value and interpretation: The behavioral signs of cognitive/psychological rejuvenation are often indirect and derived from small-scale studies or correlations. It is clear that psilocybin can have powerful and lasting psychological effects (strong evidence for antidepressant and anxiety reduction). That these effects tend towards a more youthful cognitive state (more flexibility, openness, zest for life) is a plausible interpretation, but it requires further research to specifically label this as “rejuvenation.” Nevertheless, current data suggest that psilocybin users—at least temporarily or through therapy—can become cognitively sharper and emotionally more resilient. This contributes to the idea that psilocybin does not merely combat symptoms, but the optimizes fundamental functioning of the brain and psyche in a way that corresponds to reversing certain age-related changes.
Conclusion
In summary: Psilocybin, the active ingredient of “magic” mushrooms, shows multifaceted effects in early studies on processes related to aging and cell rejuvenation. Tabularly, we can state: it promotes neural growth and plasticity (demonstrated in animals, suspected in humans); it slows cellular aging markers and preserves telomeres (demonstrated in human cell lines and mice); it reduces inflammatory factors and modulates the immune system (acutely in humans, structurally in preclinical models); and it activates “longevity genes” such as SIRT1 and increases neurotrophic factors such as BDNF. Many of these findings are preliminary and primarily based on animal models or in vitro systems, but together they form a consistent picture. The direct effects (e.g. on cells, mice) are promising and biologically explainable, while the indirect effects in humans (better cognition, mood, possible correlations with delayed aging) raise optimism but require further evidence.
The current strength of the evidence varies by aspect. Neuroplasticity and anti-inflammatory effects by psilocybin are supported by multiple studies (moderate to strong evidence). Effects on telomeres, SIRT1, and lifespan have been discovered very recently and are still based on a few studies (limited evidence, but groundbreaking in implications). Behavioral “rejuvenation” is difficult to substantiate, but the trends are encouraging (weak to moderate evidence, largely correlative or in small groups). Importantly, all these lines of research are still in development. Psilocybin research takes place primarily in the context of mental health care; the insights regarding aging are an expanding new field.
In conclusion can we state that psilocybin shows potential as a gero-protective agent – a substance that slows down aging processes – but that this has not yet been clinically proven. However, the convergence of findings (from cell to behavior) justifies further interdisciplinary research. As a follow-up, longitudinal studies in humans would be useful, in which biomarkers of aging (telomere length, inflammatory markers, cognitive function tests, epigenetic clocks) are measured before and after therapeutic psilocybin use. Until then, we must interpret the current results cautiously. Psilocybin is no “fountain of youth” – the data are preliminary and there are risks/ethical considerations associated with widespread use – but it offers a fascinating insight into how changes in consciousness and biochemistry could together contribute to a healthier aging process.
Sources: The findings discussed above are based on a selection of recent scientific publications, including cell and animal studies (e.g., Hecker et al., 2025 in NPJ Aging), clinical studies (Mason et al., 2023 on psilocybin and immune markers; Doss et al., 2021 on cognitive flexibility) and literature reviews (Haniff et al., 2024 on psilocybin, neurogenesis, and microglia). These references are linked below for further in-depth study. Each sub-conclusion should therefore be read in light of the cited sources and their experimental context. Further replication and expansion of this research will determine whether psilocybin can actually be used as senotherapeutic remedy – an exciting prospect at the intersection of neuroscience and gerontology.